Familiar materials will act differently at the smallest scales. Notably, diamonds—typically hard and brittle—grow strangely soft at the nanoscale. After years of not quite understanding why, a team of physicists finally managed to decode this behavior.
Using a custom-built electron microscope, researchers found that tiny diamonds had a relatively weak chemical bond between their surface layer and their core. Placing nanodiamonds under pressure concentrates the strain at an intermediate region between the diamond’s surface and the core. As a result, the tiny diamond doesn’t fracture but instead reacts like a flexible material, “enabling elasticity that is almost impossible in bulk diamond,” the researchers reported in a recent Physical Review X paper on the findings.
“This hidden mechanism may play a role in the elasticity of brittle materials and suggest the archetype of rigidity diamond can have its mechanical properties tuned at nanoscale,” the team, led by researchers at Zengzhou University and the Henan Academy of Sciences in China, noted in the study.
The stretchy point
The key mechanism behind this elasticity is the ratio between the number of atoms on a diamond’s surface layer as opposed to the diamond’s core. This ratio tends to be much larger in nanodiamonds, whereas bigger diamonds have a smaller surface-to-core ratio. This relationship dictates how diamonds act under pressure, Chongxin Shan, the study’s senior author and a material scientist at Zengzhou University, told New Scientist.
In nanodiamonds, the larger surface-to-core ratio results in weak bonds between the surface atoms and the inner core at what’s called the interfacial regions. This weaker area plays a “decisive role” in nanodiamonds’ elastic properties, the paper explained. Subjected to pressure, this interfacial zone literally serves as the wiggle room for the diamond, absorbing the shock instead of the surface and core atoms.
Double-checking
For the study, the researchers designed experiments to test about 100 different diamonds inside isolated vacuums. Smaller scales meant more risk of contaminants, so each diamond was baked at 212 degrees Fahrenheit (100 degrees Celsius) before the experiment. In each session, individual nanodiamonds were locked between two diamond indenters and connected to sensors that measured how squishy the diamond could get without fracturing.
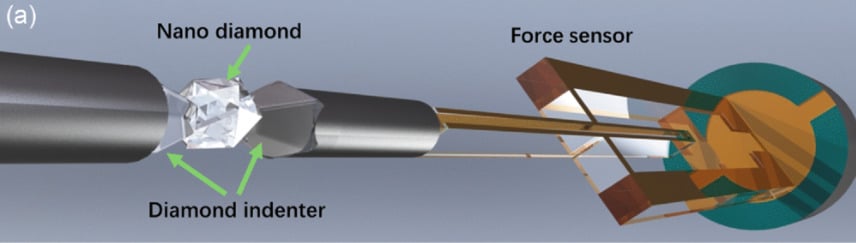
Fascinatingly, the team confirmed that size truly mattered for a nanodiamond’s elasticity. For instance, a 13-nanometer diamond had a similar rigidness as that found in common jewelry, whereas a smaller, 4-nanometer diamond was roughly 30% more stretchy. Using their observations, the researchers devised a fitted mathematical model to gauge diamond elasticity at nanoscales.
A scientist’s best friend
For scientists, diamonds are popular less for their sparkly visuals but more for their sheer versatility. To list a few examples, the capsules storing hydrogen fuel in fusion reactors are made of diamond, meaning small defects in diamond could make or break fusion reactions. Physicists are also seriously considering diamonds as tiny data storage units in quantum devices. In less futuristic applications, diamonds are already prime components of key manufacturing processes.
This idea clearly wasn’t lost on the researchers, who concluded that their findings illuminate “practical knobs for nanoscale devices, [such as] nanomechanical resonators, phononic elements, and diamond-based quantum sensors.” Assuming the latest study checks out, this could mean that researchers now have the ability to freely design more versatile, flexible components in sensitive tech.
Read the full article here











