Any concept with the term “quantum” slapped on top rarely means exactly what it’s referring to. There are always twists, turns, and conflicting interpretations that complicate things, but the answers that do arise often bring out the most fundamental truths about the universe.
In a recent Physical Review Letters paper, physicists at Goethe University in Germany take this dynamic to a whole new level, specifically for the geometry of complex atomic structures. Molecules like formic acid are thought to be “flat,” as their atoms lie on a single plane. But as the new research demonstrates, quantum mechanical effects make it so that they’re actually never flat—the tiny particles vibrate ever so slightly, ultimately giving the molecule a three-dimensional structure.
“Nature resists rigidity and forces even the simplest structures into the third dimension,” the researchers noted in a statement.
High school chemistry? Yeah, forget it
Traditional, textbook depictions of atoms and groups of atoms—molecules—typically show them as spherical objects connected with rods. This idea breaks down in the quantum world, where atomic nuclei are “more like vibrating clouds,” explained Reinhard Dörner, the study’s senior author and a physicist at Goethe University, in the statement.
To be exact, atomic nuclei are zero-dimensional “points” without a determined position at the quantum level, since they’re essentially “a little bit everywhere,” Dörner added.
Assigning shapes to probabilities
That said, scientists, for practical purposes, also often assume molecules have a fixed shape. A molecule’s geometry determines its stability and reactivity, so it makes much more sense for researchers to work with a defined model as opposed to complex wave function equations for every single reaction.
To a certain extent, scientists can identify specific angles at which atoms bind—also technically probabilities, but predictions that have gone a long way in enabling chemical breakthroughs.
When the flat thing isn’t (as) flat
The new study doesn’t reject these predictions. Rather, its findings further nuance the role of quantum mechanical effects in molecular science—a role that may have been overlooked thus far.
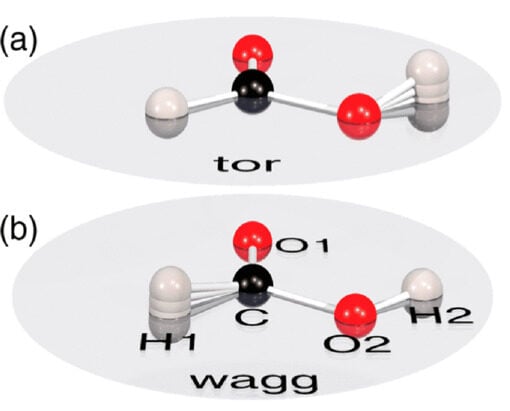
For their experiments, the researchers investigated formic acid (formula HCOOH), a colorless, smelly substance used in agricultural miticides, hydrogen storage for energy sets, chromatography, and industrial production.
The molecule is known to have a strictly planar shape, meaning its constituent atoms lie on a single, two-dimensional surface. This also makes the molecule fairly symmetrical on both sides. The researchers wanted to understand just how stable this “flat” shape really is.
Tracking the quantum trembles
To investigate, the team struck formic acid molecules with X-ray beams in a particle accelerator. When the molecule exploded from the excess energy, it released tiny electrons that the researchers used to mathematically reconstruct the original structure of formic acid in extreme detail.
Notably, they flagged consistent jiggles in two hydrogen atoms (formic acid’s formula is HCOOH) that would make the molecule “effectively three-dimensional at almost every moment,” the researchers said.
New insights into quantum molecules
Such “quantum trembling,” as the team calls it, means that formic acid likely isn’t as flat or symmetrical as was previously believed. Depending on the small vibrations of hydrogen, the molecule changes its “handedness,” or whether it slants toward the right or the left.
In chemistry, this asymmetry can change the properties of a molecule altogether. That said, the researchers don’t believe this calls for a conceptual overhaul; the widespread use of formic acid itself serves as proof that scientists are well-versed in its behavior.
Instead, quantum trembling suggests that handedness might not be as dependent on a static molecule’s geometry as was previously believed.
“As we were able to show using the example of formic acid, quantum trembling alone can generate two different mirror-image realities from a symmetrical molecule,” Dörner said. “Our findings with formic acid show that geometry is not a static property but a dynamic event.”
The study also exemplifies how quantum effects always seem to complicate what we think we know about things at the macroscopic level. These quantum effects are very real. So is our macroscopic reality. But the discrepancies between the two somehow don’t break down our reality—the reality we live in every day.
Read the full article here